
What's going on here? Obviously, the lake is not boiling, and - obviously - there isn't a general fog blanketing the entire forest, so why does the lake look like a pot on the stove? Well, it's due to many of the same principles of what happens when water boils in a pot on the stove, namely evaporation and condensation, but you have to think of these things in terms of heat energy and NOT in terms of water temperature alone.
In the case of boiling water on a stove, water is heated, causing evaporation; the gaseous water requires a certain amount of heat energy to maintain its gaseous state, and if the heat energy of the gaseous water falls below the condensation point, it turns back into liquid water, forming a fine mist. The mist increases in density as the amount of condensing water increases. This is why you see more and more steam rising from a pot of water as it reaches boiling point.
However, it's not only the temperature of the water that you have to consider when thinking about steam formation. In addition to the process of condensation described above, there is also the humidity to consider. In other words, the air itself can only "hold" a certain amount of gaseous water, and this capacity is determined by the temperature of the air; the higher the air temperature, the more water can be held as a gas. This is why there's a lot more steam seen above a pot boiling in very humid conditions than in very dry conditions (even when the air temperatures are identical).
In the case of misty lakes in the morning, all these processes are happening, just like in the example of the boiling pot, except the temperatures are far lower. Throughout the summer, the lake has absorbed and retained a large amount of heat energy in the top layer of the lake. At night -- especially on cloudless nights -- the heat energy in the air rises away from the surface and escapes this local system, thus bringing down the temperature. There is evaporation taking place in the lake, both day and night, thanks to the higher temperatures of the surface of the lake. However, unlike during the heat of the day, at night, since there is a lower temperature, there is less capacity for the air to hold on to gaseous water, and we can measure this as an increase in humidity. As the humidity rises to 100%, the air has a diminishing capacity to hold on to all the water that is evaporating -- due to the heat of the water in the lake -- and so the water almost immediately condenses into mist as it rises from the lake. In fact, if you heat a pot to the same temperature of the lake water, you would see steam flowing up from your pot, even though the water temperature is nowhere near boiling.
Once the sun's rays strike the surface of the water, the humidity is "burned away", in that the local temperature increases enough so that the humidity drops below 100%, thus allowing the air to once again absorb the evaporating water.
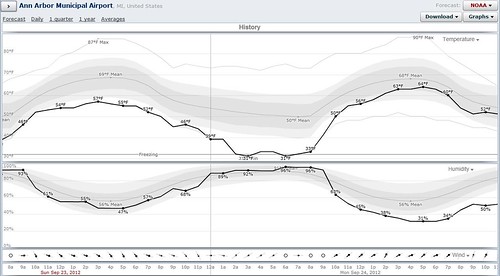
Of course, the reason why the lake effectively billows with steam during early fall mornings is due to the relatively large amount of heat stored in the lake combined with one or two significantly cold mornings. The photo above was taken on September 24, 2012, and if we look at the weather conditions measured at the Ann Arbor airport (about five miles away), we see that the overnight temperature dropped to freezing, and the humidity was near saturation. (Of course, this was in an open field, not above a lake, where the saturation would have been 100%.)
Yeah, okay, so this was a bit of a rambling post, but I hope that it helped you understand (if you didn't already) why so much mist rises off the lake on cool autumnal mornings. Of course, there is little reason to necessarily understand the why in order to appreciate the effect. In the end, fall is a veritable feast of sights, just like spring was one of smells, and summer one of sounds.
No comments:
Post a Comment